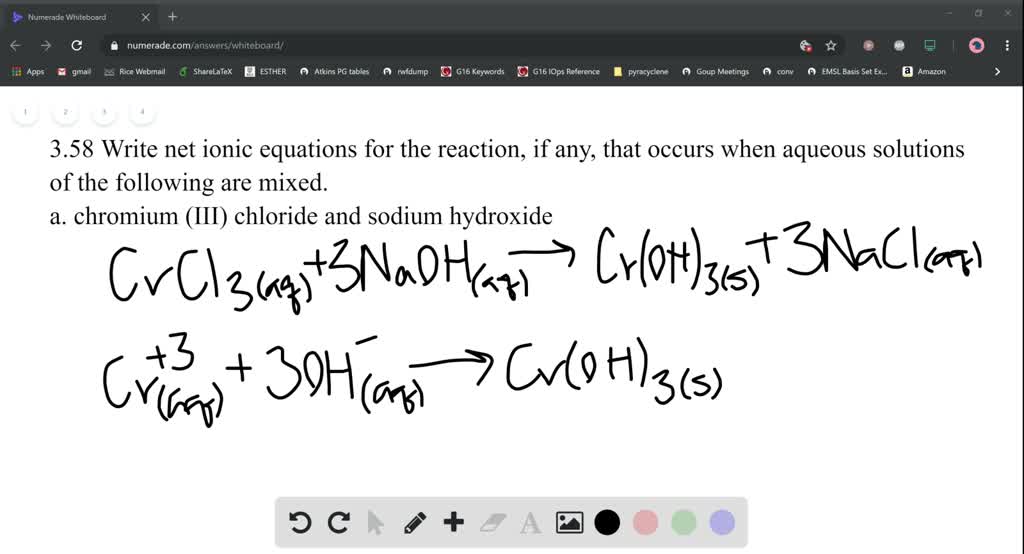
Sulfur dioxide is bubbled into the solution until. Neither ILO nor WHO nor the European Commission shall be responsible for the interpretation and use of the information contained in this material. Twenty-five grams of potassium dichromate (or 17g of chromium(VI) oxide) are dissolved in 500ml of water. The published material is being distributed without warranty of any kind, either expressed or implied. When chromium (III) chloride is combined with sodium hydroxide, it forms chromium (III) hydroxide (a colourful green precipitate). 
The molecular weight may vary depending on the degree of hydration.Īll rights reserved. The name Chromic acid is used for this product, as well as for Chromic (VI) acid (CAS ) and for Chromium (VI) oxide (CAS, see ICSC 1194). Visit ChemicalBook To find more CHROMIUM(III) ACETATE HYDROXIDE(3) information like chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight, physical properties,toxicity information,customs codes. TLV: (as Cr(III), inhalable fraction): 0.003 mg/m 3, as TWA (RSEN) (DSEN) A4 (not classifiable as a human carcinogen). May cause mechanical irritation to the eyes.Įvaporation at 20☌ is negligible a nuisance-causing concentration of airborne particles can, however, be reached quickly, especially if powdered.Įffects of long-term or repeated exposure Uses advised against Food, drug, pesticide or biocidal product use. : 12518 CAS-No 3 Synonyms No information available Recommended Use Laboratory chemicals. The substance can be absorbed into the body by inhalation and by ingestion. Product Name Chromium(III) acetate hydroxide Cat No. With the financial assistance of the European Commission.ĭecomposes on heating. Prepared by an international group of experts on behalf of ILO and WHO, Chromium (III) hydroxide trihydrate CrH12O96915 - structure, chemical names, physical and chemical properties, classification, patents. Then store and dispose of according to local regulations. If appropriate, moisten first to prevent dusting. Sweep spilled substance into covered containers. Personal protection: particulate filter respirator adapted to the airborne concentration of the substance. Rinse skin with plenty of water or shower.įirst rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention. In case of fire in the surroundings, use appropriate extinguishing media.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
